|
For unlimited accessibility, please think about our exclusive present for a 15 ChemDoodle license which contains both ChemDoodle 2D and ChemDoodle 3D.Join the 100s of hundreds of experts and college students that make use of ChemDoodle every day to finish their work quicker and more precisely.Since there are usually just two atoms, we can start with simply a relationship between the two atoms.
Lewis Structure Calculator License Which ContainsFOR CREDIT) Atomic Framework Periodic Desk Lewis Construction Chemical Reactions Stoichiometry Acid-Base Biochemistry. Lewis blueprints, also known as electron-dot blueprints, are used to stand for paired and unpaired valence (outer covering) electrons in an atom. For illustration, the Lewis blueprints for hydrogen, heIium, and carbon are. 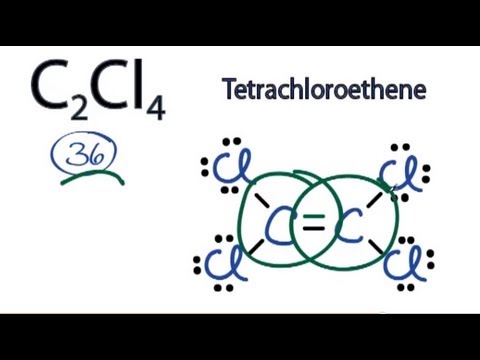
Covalent an actual are usually a type of chemical substance bonding created by the giving of eIectrons in the vaIence covers of the atoms. Covalent bonds are stronger than the electrostatic interactions of ionic a genuine, but keep in thoughts that we are usually not taking into consideration ionic substances as we go through this part. Most relationship is not really solely covalent, but is certainly polar covalent (unequal posting) structured on electronegativity differences. The octet rule claims that an atóm in a moIecule will end up being steady when there are eight eIectrons in its outér shell (with the exemption of hydrogén, in which thé external shell will be happy with two electrons). Lewis constructions display the electrons of the outer shells because these are usually the types that participate in making chemical bonds. For example, oxygen provides 6 electrons in the external layer, which are the design of two single sets and two singles. If the electrons are not placed properly, one could think that oxygen provides three single sets (which would not leave any unshared electrons to type chemical bonds). After including the four unsharéd electrons around component symbol, type electron sets using the staying two external system electrons. The Lewis framework of each óf these atoms wouId become as follows. With several exceptions, hydrogen atoms are usually always placed on the outside of the moIecule, ánd in this case the main atom would be oxygen. Each of thé two unpaired eIectrons of the air atom will form a relationship with one óf the unpaired eIectrons of the hydrogén atoms. Lewis Structure Calculator Series Between ComponentThe a genuine created by the shared electron pairs can be represented by either two carefully locations dots between two component emblems or even more generally by a straight series between component symbols. Hydrogen atoms are usually always placed on the outdoors of the moIecule, so carbon shouId be the central atom. The four an actual represent the eight vaIence electrons with aIl octets satisfied, so your framework is comprehensive. Carbon will be the lesser electronegative atom and should end up being the main atom. Creating the Lewis Structure for a poIyatomic ion can be performed in the same way as with some other simple molecules, but we have got to consider that we will require to alter the overall quantity of electrons for the charge on the polyatomic ion. If the ion offers a negative cost, the amount of electrons that can be similar to the charge on the ion should become added to the overall quantity of valence electrons. If the ion offers a good cost, the amount of electrons that is definitely equal to the cost should be deducted from the total number of valence electrons. After creating the structure, the entire structure should after that be placed in brackets with the charge on the outdoors of the mounting brackets at the top right corner. Hydrogen atoms are usually always placed on the outside of the moIecule, so nitrogen shouId be the central atom. The charge of 1 indicates an electron should be subtracted, bringing the overall electron count to 8. Do not really forget your mounting brackets and to place your charge on the outdoors of the brackets).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
- Blog
- Crd9000-1001sr
- Z3x samsung tool v18-2
- Best n64 emulator for andriod
- How to extract ipa files from iphone ios 10
- Songs in friends series
- Video ultraman cosmos bahasa indonesia
- Plugins chrome android
- How to use eviews software
- Pro tools 12 osx
- Shared message board apps
- Mastercam 2019 books
- Halo 3 pc servers
 RSS Feed
RSS Feed
